Maths-
General
Easy
Question
- 6
- 1

- 0

It is sequence, it can be seen easily that it tends to 0.In this question, we have to find value of  .
.
The correct answer is: 




On substituting, We get

Related Questions to study
physics-
The figure shows P–V diagram of a thermodynamic cycle If  are the respective temperature at A, B, C and D Then, choose the correct statement if
are the respective temperature at A, B, C and D Then, choose the correct statement if 

The figure shows P–V diagram of a thermodynamic cycle If  are the respective temperature at A, B, C and D Then, choose the correct statement if
are the respective temperature at A, B, C and D Then, choose the correct statement if 

physics-General
physics-
The figure shows P–V diagram of a thermodynamic cycle The work done by the cycle is

The figure shows P–V diagram of a thermodynamic cycle The work done by the cycle is

physics-General
physics-
A monoatomic ideal gas sample is given heat Q One fourth of this heat is used as work done by the gas and rest is used for increasing its internal energy An ideal gas under goes a thermodynamic cycle as shown in fig Which of the following graphs represents the same cycle?
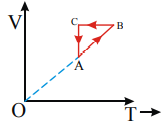
A) 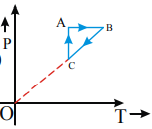
B) 
C) 
D) 
A monoatomic ideal gas sample is given heat Q One fourth of this heat is used as work done by the gas and rest is used for increasing its internal energy An ideal gas under goes a thermodynamic cycle as shown in fig Which of the following graphs represents the same cycle?

A) 
B) 
C) 
D) 
physics-General
physics-
A monoatomic ideal gas sample is given heat Q One fourth of this heat is used as work done by the gas and rest is used for increasing its internal energy The P V diagram for the process is
A monoatomic ideal gas sample is given heat Q One fourth of this heat is used as work done by the gas and rest is used for increasing its internal energy The P V diagram for the process is
physics-General
physics-
The V–T diagram of an ideal gas for the process  (straight lines) is as shown in the figure In the process
(straight lines) is as shown in the figure In the process 
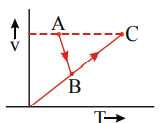
A) pressure is always increasing
B) for some interval pressure decreases but finally pressure is more than initial pressure
C) pressure first increases then remains constant
D) graph AB is unpredictable about pressure
The V–T diagram of an ideal gas for the process  (straight lines) is as shown in the figure In the process
(straight lines) is as shown in the figure In the process 

A) pressure is always increasing
B) for some interval pressure decreases but finally pressure is more than initial pressure
C) pressure first increases then remains constant
D) graph AB is unpredictable about pressure
physics-General
physics-
Statement-1: Two vessels A and B are connected to each other by a stopcock .Vessel A contains a gas at 300K and 1 atmosphere pressure and vessel B is evacuated The two vessels are thermally insulated from the surroundings If the stopcock is suddenly opened, the expanding gas does no work
Statement-2: Since D Q = 0 and as the gas expands freely so DW = 0 and from the first law of thermodynamics it follows that DU is also zero for the above process
Statement-1: Two vessels A and B are connected to each other by a stopcock .Vessel A contains a gas at 300K and 1 atmosphere pressure and vessel B is evacuated The two vessels are thermally insulated from the surroundings If the stopcock is suddenly opened, the expanding gas does no work
Statement-2: Since D Q = 0 and as the gas expands freely so DW = 0 and from the first law of thermodynamics it follows that DU is also zero for the above process
physics-General
Maths-
We can only apply the L’Hospital’s rule if the direct substitution returns an indeterminate form, that means 0 over 0 or
Maths-General
We can only apply the L’Hospital’s rule if the direct substitution returns an indeterminate form, that means 0 over 0 or
physics-
A 5 m long alummininm wire  of diameter 3 men supports a 40 kg mass. In order to have the same elongation in a copper wire
of diameter 3 men supports a 40 kg mass. In order to have the same elongation in a copper wire  of the same length under the same weight, the diameter should now be in men.
of the same length under the same weight, the diameter should now be in men.
A 5 m long alummininm wire  of diameter 3 men supports a 40 kg mass. In order to have the same elongation in a copper wire
of diameter 3 men supports a 40 kg mass. In order to have the same elongation in a copper wire  of the same length under the same weight, the diameter should now be in men.
of the same length under the same weight, the diameter should now be in men.
physics-General
physics-
Statement-1: First law of thermodynamics specifies the conditions under which a body can use its heat energy to produce the work
Statement-2: Second law of thermodynamics states that heat always flows from hot body to cold body by itself
Statement-1: First law of thermodynamics specifies the conditions under which a body can use its heat energy to produce the work
Statement-2: Second law of thermodynamics states that heat always flows from hot body to cold body by itself
physics-General
maths-
A random variable has the following distribution  Then for the values, A = K, B = Mean,
Then for the values, A = K, B = Mean,
C = Variance, the ascending order is
A random variable has the following distribution  Then for the values, A = K, B = Mean,
Then for the values, A = K, B = Mean,
C = Variance, the ascending order is
maths-General
Maths-
The probability distribution of a random variable X is given below  A:p (2 ≤ x < 4) B:p(x≥ 4) C p (x ≤ 3) D:p(3≤ x≤ 5) Arrange A, B, C, D in ascending order of magnitude
A:p (2 ≤ x < 4) B:p(x≥ 4) C p (x ≤ 3) D:p(3≤ x≤ 5) Arrange A, B, C, D in ascending order of magnitude
The probability distribution of a random variable X is given below  A:p (2 ≤ x < 4) B:p(x≥ 4) C p (x ≤ 3) D:p(3≤ x≤ 5) Arrange A, B, C, D in ascending order of magnitude
A:p (2 ≤ x < 4) B:p(x≥ 4) C p (x ≤ 3) D:p(3≤ x≤ 5) Arrange A, B, C, D in ascending order of magnitude
Maths-General
maths-
A random variable X has the probability Distribution  Events E= {X is a prime number} and F= {X / X < 4}
Events E= {X is a prime number} and F= {X / X < 4}


Which of I, II is (are) true
A random variable X has the probability Distribution  Events E= {X is a prime number} and F= {X / X < 4}
Events E= {X is a prime number} and F= {X / X < 4}


Which of I, II is (are) true
maths-General
Maths-
A random variable x has the following probability distribution  Determine P(X > 6)
Determine P(X > 6)
A random variable x has the following probability distribution  Determine P(X > 6)
Determine P(X > 6)
Maths-General
Maths-
The range of a random variable X is {0,1, 2} and  , P(x = 2)= 5K -1. Then we have
, P(x = 2)= 5K -1. Then we have
The range of a random variable X is {0,1, 2} and  , P(x = 2)= 5K -1. Then we have
, P(x = 2)= 5K -1. Then we have
Maths-General
Maths-
If the range of random variable  and
and  for k =0,1, 2,.....then c =
for k =0,1, 2,.....then c =
If the range of random variable  and
and  for k =0,1, 2,.....then c =
for k =0,1, 2,.....then c =
Maths-General



